Background
Natural products have historically been an invaluable source of therapeutic agents. Compounds from biological sources continue to play an extremely important role in the development of therapeutics mainly because of their enormous structural diversity, serving as privileged scaffolds in drug discovery. In fact, almost half of the drugs introduced between 1940 and 2012 were of natural origin or inspired by natural compounds, and natural products clearly have a most dramatic impact in the area of cancer.
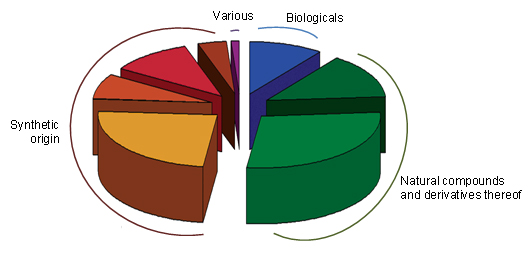
All available anticancer drugs, 1940s-2012, by source.
Natural compounds or derivates: green, synthetic origin: red/yellow, biologicals: blue, various: purple.
Besides their importance as scaffolds for drug design, natural products have also proven to be valuable and indispensable tools for deciphering cellular processes and identifying targets in the field of signal transduction research. For example Elucidation of the mechanism of action of rapamycin, which is produced by a strain of Streptomyces hygroscopicus, led to the identification of a kinase subsequently named mTOR (mammalian target of rapamycin).
Despite its potential, natural product research was de-emphasized in large parts of the pharmaceutical industry in the 1990s and considered unable to compete with the efficiency of combinatorial chemical library synthesis and screening. This was mainly due to difficulties in isolation and characterization of active compounds (via high-throughput screening) and the complexity of resupply. Moreover, difficulties associated with natural product synthesis and semi-synthesis present an obstacle to their acceptance as leads for optimization. On the other hand the pharmaceutical industry has been suffering from a lack of productivity in terms of the number of new chemical entities approved. This can at least partly be attributed to insufficient quality and quantity of lead structures.
However, because of their rich functionality providing potency and selectivity, there is clearly an “evolving role” for natural products in drug discovery and “natural products” are without doubt both of high scientific relevance as well as of high actuality. To this end, strategies to overcome major obstacles in natural product development, such as resupply/ synthesis as well as straightforward testing of functional relevance are needed.
This Research Group focuses on myxobacteria as a highly attractive source for innovative compounds, which will then be used as chemical tools and lead structures in the field of cancer therapy.
Myxobacteria
These Gram-negative bacteria are appreciated for their extraordinary ability to produce secondary metabolites with a high degree of structural biodiversity and distinct biological (including anti-tumor) activity. Epothilones targeting microtubules are the most prominent example. A semi-synthetic epothilone analog, ixabepilone, has recently been approved by the US Food and Drug Administration for metastatic breast cancer.
Innovative biotechnological/(bio)synthetic approaches can be taken using this group of microorganisms to guarantee validated supply. Moreover, the construction of novel compound analogs is considerably facilitated by the application of biosynthetic engineering and its combination with powerful chemical synthesis, so called “mutasynthesis”, based on the feeding of elaborate precursor molecules to this microorganism.
Test Systems
The focus on myxobacterial compounds serving as chemical tools and therapeutic lead structures calls for “meaningful test systems for bioactivity”.
Our attention is paid on high content cellular assays extended by in vivo proofs of anticancer activity. In contrast to single isolated target screening, such as isolated cell surface receptors or enzymes as usually applied in industrialized high throughput screening (HTS), cellular testing allows the identification of the biological functionality of compounds. Our test systems for anti-tumor and anti-migratory activity will focus
- on various types of cells involved in tumor progression and metastasis such as highly invasive tumor cells (pancreatic, mammary, hepatoma, colonic tumor cell lines), endothelial cells (angiogenesis), and immune cells (leukocytes), and will focus
- on functionally important processes, namely cell migration, cell metabolism, cancer immune-surveillance and apoptosis induction.
- on efficacy in more complex systems will be obtained by employing tumor cell spheroids and in vivo mouse tumor models.
There are several rationales for this design of functional testing:
- Complex processes of tumor, and metastasis formation depend not only on tumor cells, but also involve the tumor microenvironment.
- Inflammation and motility as well as metabolism of tumor cells and tumor endothelial cells are hallmarks of tumor development, metastasis, and tumor angiogenesis. The same is true for resistance of tumor cells to apoptosis. Myxobacteria-derived products often target the eukaryotic cytoskeleton and are thus likely to affect motility and adhesion processes.
- Tumor cell 3D- as well as in vivo testing has to be accomplished at a rather early stage of compound characterization.
